
Thus, this experiment clearly verifies the law of conservation of mass. The mass of the tube is found to be exactly the same as the mass obtained before inverting the tube. The reaction takes place and a white precipitate of silver chloride is obtained. 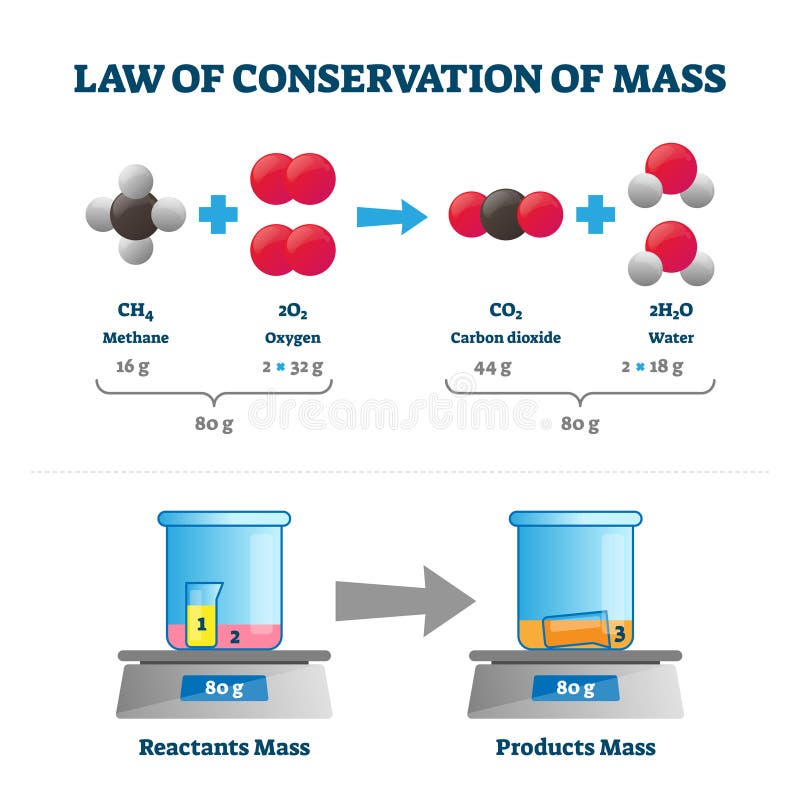
Now the tubes is averted so that the solutions can mix up together and react chemically. Sodium chloride solution is taken in one limb of the tube and silver nitrate solution in the other limb as shown in figure.īoth the limbs are now sealed and weighed. Procedure: A specially designed H-shaped tube is taken. This means the total mass of reactants in a chemical reaction will equal the total mass of the products. Requirements: H-shaped tube called Landolt’s tube, Sodium chloride solution, silver nitrate solution, etc. The law of conservation of mass indicates that mass cannot be created nor destroyed. A concise way of expressing this law is to say that the amount of matter in a system is. It states that in any given system that is closed to the transfer of matter (in and out), the amount of matter in the system stays constant. 
Famous scientists did not always agree on God and his dice but they were not. One scientific law that provides the foundation for understanding in chemistry is the law of conservation of matter. Science and religion are human knowledge. However it may change from one form to another. Energy is eternal according to the scientific law of conservation of energy. During any change (physical or chemical), matter is neither created nor destroyed. Law of conservation of mass: It states that mass can neither be created nor destroyed in a chemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
